Ionic radius9/28/2023   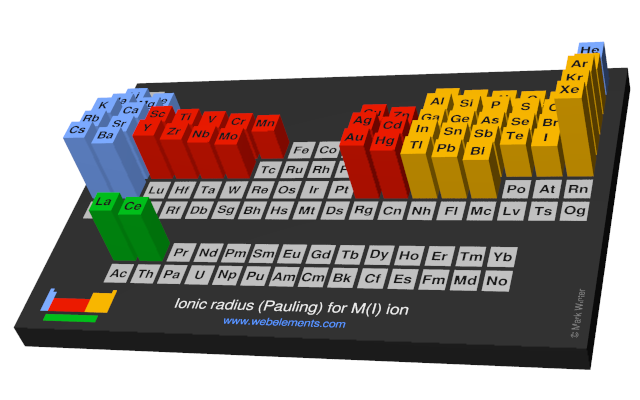
From geometric considerations, the anion radius in this case is given by:Īnd thus the radii of the S 2- and Se 2- ions are 1.84 and 1.93 Å, respectively. This suggests that the anions are in contact in these structures. All of these compounds crystallize in the NaCl structure:įor the two larger anions (S 2- and Se 2 -), the unit cell dimensions are the same for both cations. Consider for example the compounds MgX and MnX, where X = O, S, Se. We can build up a table of ionic radii by assuming that the bond length is the sum of the radii (r + + r -) if the ions are in contact in the crystal. Ionic radii depend on oxidation state (higher charge => smaller cation size, larger anion size).Bond distances (and therefore ionic radii) depend on bond strength and coordination number (remember Pauling's rule D(n) = D(1) - 0.6 log n).Atoms in crystals are not really ions - there is a varying degree of covalency depending electronegativity differences.We don't know the radii of individual ions.There are unfortunately several challenges with using this idea to predict crystal structures: The stability limit is at r C/r A = 0.414 This diagram is for coordination number six: 4 anions in the plane are shown, 1 is above the plane and 1 is below. The table below gives the ranges of cation/anion radius ratios that give the best fit for a given coordination geometry.Ĭritical Radius Ratio. If the cation is too small for a given site, that coordination number will be unstable and it will prefer a lower coordination structure. If the cation can touch all of its nearest neighbor anions, as shown at the right for a small cation in contact with larger anions, then the fit is good. Cations will find arrangements in which they can contact the largest number of anions. We consider that the anion is the packing atom in the crystal and the smaller cation fills interstitial sites ("holes"). The basic idea of radius ratio rules is illustrated at the right. Going from left to right across any row of the periodic table, neutral atoms and cations contract in size because of increasing nuclear charge. Note that cations are always smaller than the neutral atom (pink) of the same element, whereas anions are larger. Pauling proposed from geometric considerations that the quality of the "fit" depended on the radius ratio of the anion and the cation.Ītomic and Ionic Radii. The first such rules were proposed by Linus Pauling, who considered how one might pack together oppositely charged spheres of different radii. It was (and still is!) important to develop some guidelines for guessing the coordination numbers and bonding geometries of atoms in crystals. Once a trial structure was proposed, it was relatively easy to calculate the diffraction pattern, but it was difficult to go the other way (from the diffraction pattern to the structure) if nothing was known a priori about the arrangement of atoms in the unit cell. 
The covalent radius of Cl 2 is half the distance between the two chlorine atoms in a single molecule of Cl 2.\)Įarly crystallographers had trouble solving the structures of inorganic solids using X-ray diffraction because some of the mathematical tools for analyzing the data had not yet been developed. (d) This is a depiction of covalent versus van der Waals radii of chlorine. (c) The van der Waals atomic radius, r vdW, is half the distance between the nuclei of two like atoms, such as argon, that are closely packed but not bonded. (b) The metallic atomic radius, r met, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as aluminum. (a) The covalent atomic radius, r cov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
